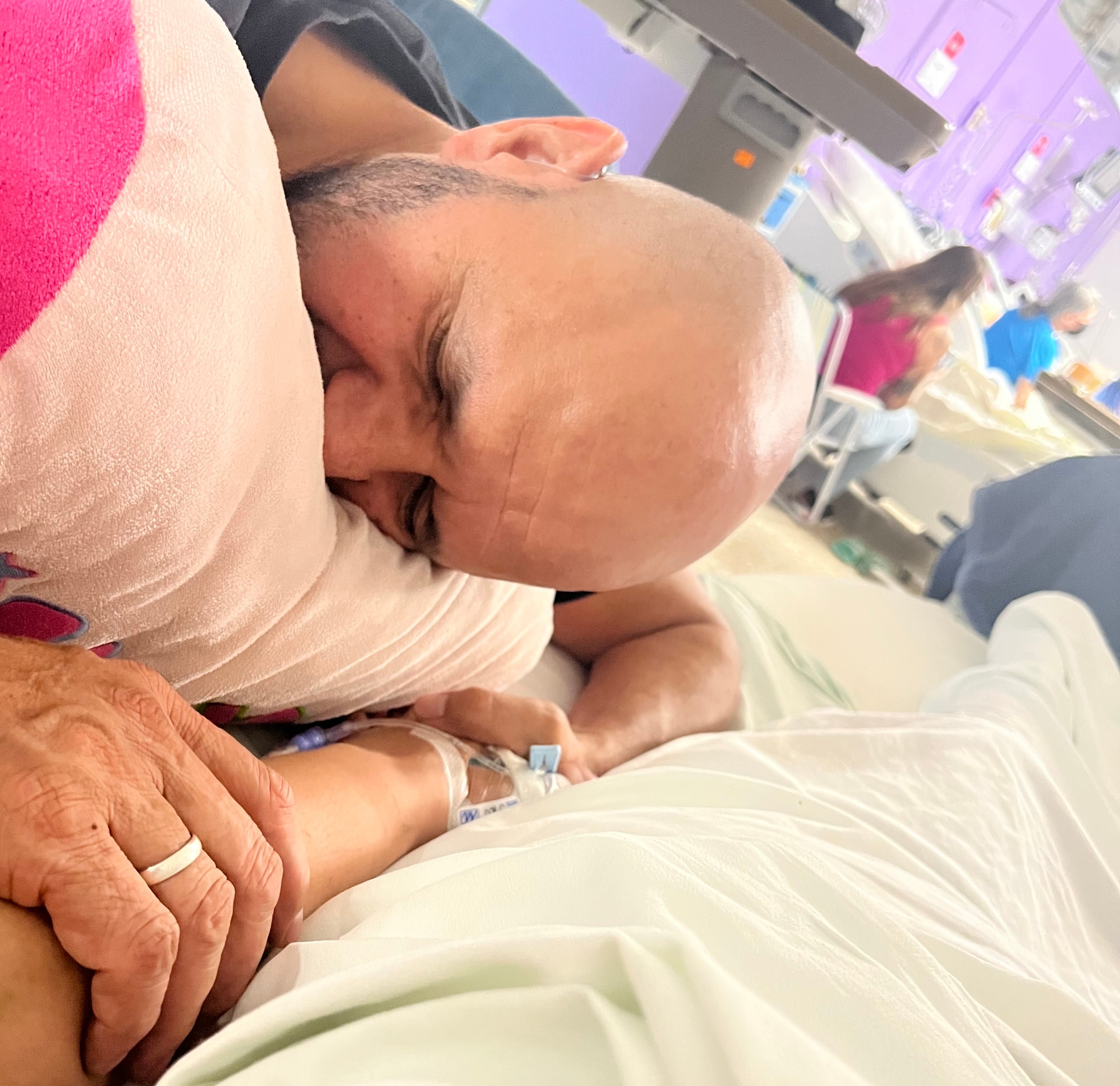
Andrea Picado Zamora was 33 years old when she was diagnosed with triple-negative breast cancer, a subtype with options for reduced treatment.
This Special Education teacher and resident of Calles Fallas, Desamparados, received all the cycles of chemotherapy that her oncologist prescribed; She then underwent a radical mastectomy of the right breast and, finally, radiation therapy.
When the standard protocol was finished, the doctor prescribed pembrolizumabwhose trade name is Keytrudaan oncology drug that helps the immune system identify and destroy cancer cells.
However, the Costa Rican Social Security Fund (CCSS) denied it. The reason was the cost of the drug, according to the paper that the entity delivered.
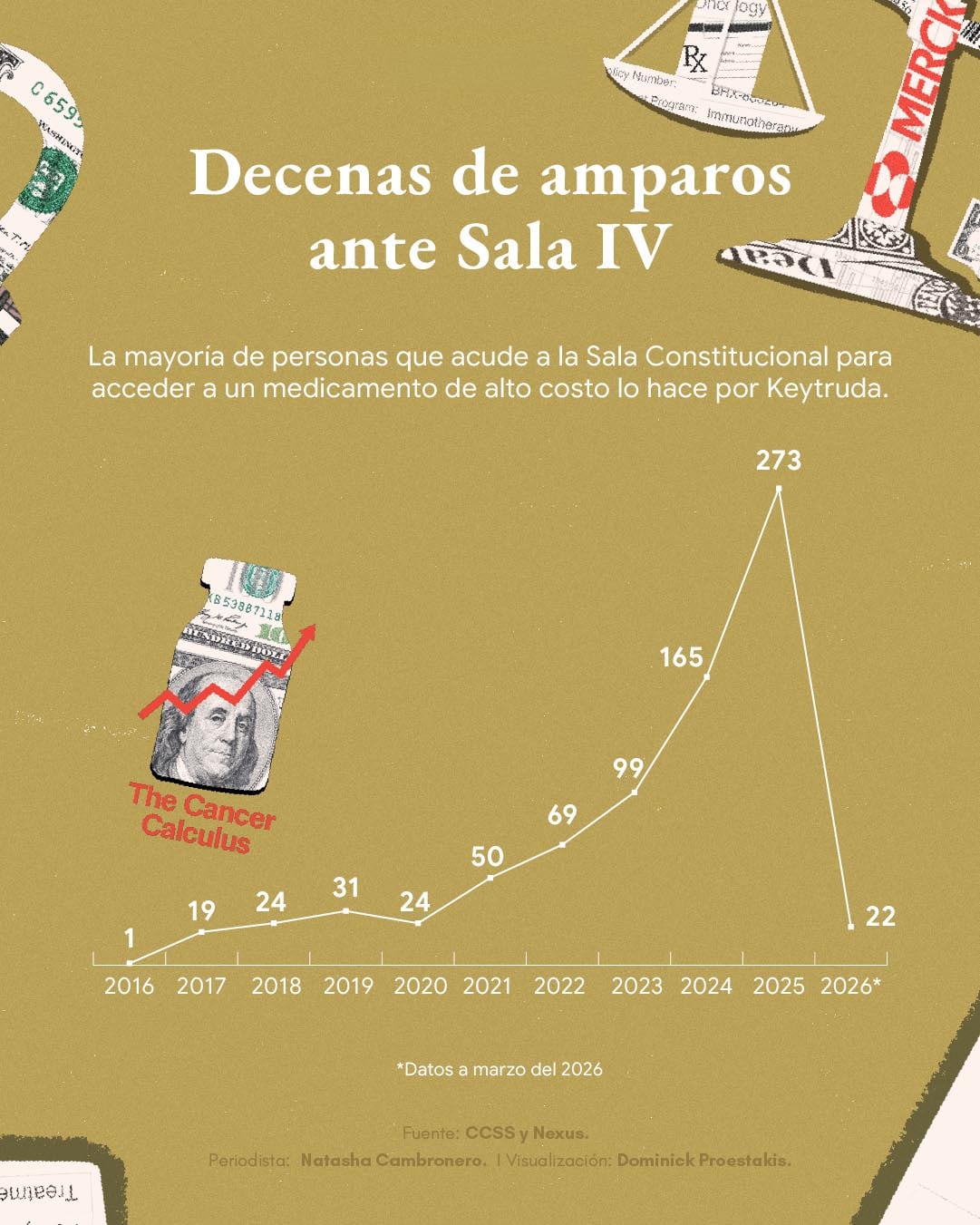
This is the medicine where the CCSS spends the most money and for which more protection resources receives from patients, who go to the Constitutional Chamber to claim treatment.
two years ago, Each dose of this medication cost the Fund $2,900 and at the private level it was around $5,000.. For this reason, Social Security does not prescribe it to all patients, while patients cannot purchase it on their own.
There are also no options to search for a similar drug. This immunotherapy is only produced and marketed by the pharmaceutical company Merck & Co. (MSD), which has its patent protected at least until 2028.
Now, after a two-year negotiation—declared confidential—the CCSS achieved a reduction and is purchasing this medication for $1,440 per dose, 49.6% less, which will allow it to be supplied to patients with 21 types of cancer tumors.
This report is part of the global investigation, The million dollar medicine (The Cancer Calculus)coordinated by the International Consortium of Investigative Journalists (ICIJ).
The Via Crucis of the Picado family
Given the refusal, his father, Víctor Picado, spent almost an entire month going to the San Juan de Dios Hospital to look for directors, pharmacologists and officials.
Their goal was for the medical center to will deliver the formal response to the rejection to be able to file an appeal for protection.
“In fact, everyone in San Juan de Dios already knows him,” joked Andrea, now 36 years old.
The document—a letter that the institution does not issue automatically and that the patient must request at the counter—took about a month to arrive.
Briceida Cantillo Hernández, president of the Second Chance at Life National Association (Anasovi)an organization that since 2011 has filed appeals for cancer patients, described this wait as one of the most absurd aspects of the process and difficult for patients and families.
“Sometimes it takes from 22 days to a month to respond, and it is only to make a photocopy for the patient,” Cantillo explained. “In all this, at least one quarter is lost,” he added.
Protection as the only way
With the letter in hand, the Picado family approached Anasovi and Andrea managed to file an appeal for protection before the Constitutional Court. And he won. “I think I was lucky, really,” he stressed.
The entire process—from the formal rejection of the CCSS to the first dose—took him almost two months and 21 days: a month waiting for the written rejection that his father sought without pause, another month for the protection to be declared valid, and 21 more days until he received the first dose.
In 2024 alone, the Fund received 648 appeals for high-cost medications and, of that number, 165 (25%) were for pembrolizumab (Keytruda).
In second place was nintedanib – which is used to treat pulmonary fibrosis – with 48 protections, that is, 7.4%. The remaining 435 resources were for another 54 medications, according to data provided by Social Security.
Last year, the trend was maintained. On the Nexus platform, for jurisprudence of the Judiciary, 273 rulings from the Constitutional Chamber linked to Keytruda appear. And in the first three months of 2026, there are 22.
An almost tacit bureaucracy
He Dr. Larry Ramírez Chavarría, spokesperson for the College of Pharmacists of Costa Ricaexplained that this route has become a almost tacit bureaucracy: The Central Pharmacotherapy Committee rejects the request and the patient has the protection action ready to file almost in parallel, because the rejection is foreseeable.
“The problem is that the Chamber is not a technical entity from a medical point of view. What can a magistrate do to determine if the treatment is appropriate? They have to trust what the treating doctor says. And meanwhile, the person spends months in that process”Ramirez said.
The Central Committee for Pharmacotherapy (CCF) is the permanent advisory body of the CCSS that prepares the Official List of Medicines and authorizes whether a drug is delivered or not. It meets only once a week.
Their frequent rejections are justified by the argument that the available treatments have not been exhausted, and by the weight of cost on institutional sustainability.
However, Dr. Ramírez acknowledged that the filter is necessary—there are expensive treatments that do not offer real differences in survival—but he questioned that the committee does not weigh carefully sufficient weight the patient’s quality of life.
The procedure
When a treating physician requests the medication, the patient must refer it to the local pharmacotherapy committee of the hospital where he or she is treated. There the case is evaluated and then raised to the CCF, whose members meet once a week.
If the cases are not reviewed in that session, they are left for the next one. If the session falls on a holiday, it is postponed for another week.
“The patient doesn’t have time; that’s what I mean”stressed Briceida Cantillo, who assured that she has had cases of patients who, with the appeal declared valid, die in the bureaucratic process before the first dose.
“If the Central Committee does not speed up, the cases accumulate. Then more days pass until the hospital responds. If the patient is not attentive or lives outside the Greater Metropolitan Area, they find out at the next appointment. Thus a month, two months pass to discover that the medication was rejected,” explained the president of Anasovi.
Even when Chamber IV orders delivery, the process does not end. The case goes to the Goods and Services Acquisitions Area of the CCSS, which Cantillo described as “the worst hardship that a patient has in accessing medicine.”
The institution has 15 days to deliver. If they do not comply, the patient must submit a request for compliance with the sentence. Then he poses a second one. The third request implies legal consequences for officials.
“Magically, the medication appears in the patient’s hands, but how long did the patient go without it?” Cantillo asked.
The answer, in at least one case that Anasovi’s president documented, was death.
It was about a woman with kidney cancer who won the appeal for protection when she was still in good health. She died before receiving the approved medication.
“I have photos of her, how well that lady was when she won the appeal,” he said. “During this entire period of time, the patient died without receiving it”he added.
The real cost of pembrolizumab
The medication in questionpembrolizumab, marketed as Keytruda— is a monoclonal antibody.
In the CCSS, a 25 mg/ml bottle has cost up to $2,900; in the private sector, up to $5,000. It is not a fixed-cycle treatment: the oncologist evaluates the response of the tumor and decides whether to continue, modify the dose or add another component. It can go on for months.
Oncologist Allan Ramos Esquivel, professor of Pharmacology at the University of Costa Rica and head of the Medical Oncology service at the San Juan de Dios Hospital, also questioned its difficult access.
“Almost ten years have passed since its commercialization and today its safety and effectiveness are known in more detail. Decisions that were previously taken into account in this process are now a thing of the past,” said Ramos.
However, he acknowledged that the underlying problem persists: “The cost is very high for any country, and much more so for Costa Rica. Hence the need to negotiate with the industries and propose payment alternatives.”
‘I decided to fight as far as God wants’
Andrea Picado received pembrolizumab for a full year, from June 2023 to June 2024. Today she continues another treatment with a different medication. Eat better. Walk better.
When he talks about the diagnosis and everything that came after—the chemotherapy, the surgery, the months of negotiations, the uncertainty—his voice does not exude bitterness.
“I think most people when they say cancer, hey ‘death‘. But I am very spiritual, I have trusted God a lot since I was little. I decided to fight as far as God wants. It has been distressing, I am not going to say no, painfuland it is that uncertainty of whether tomorrow I will wake up alive, but all that peace, all that strength that I can transmit, comes from God,” he said.
She also mentioned the network that supported her: family, cousins, uncles, parents, sisters, nephews and friends. “I never imagined I would have a support network of that size,” he said.
His conclusion about cancer is brief and does not allow for nuances: “You have to grab it by the horns and move forward.”.