After two years of negotiations and a confidential agreement, the Costa Rican Social Security Fund (CCSS) achieved a 49.6% reduction in it medication you spend the most money on and for which more protection resources receives from patients, who go to the Constitutional Chamber to claim treatment.
It is about the pembrolizumab, brand name Keytrudaa immunotherapy oncology drugwhich is used to treat conditions such as:
- Melanoma
- kidney cancer
- head cancer
- Neck cancer
- breast cancer
- Hodgkin lymphoma
- Cervix cancer
This drug is only produced and marketed by the pharmaceutical company Merck & Co. (MSD) and his patent is protected, at least, until 2028which prevents another company from generating a biosimilar to compete with it and make it cheaper.
Unlike chemotherapy, which damages healthy tissue while destroying cancer cells, immunotherapy treatment helps the patient’s system so that identify and destroy them.
By having a single bidder, the pharmaceutical company sets the sales price and, therefore, the CCSS saw the need to negotiate.
In 2024, the Fund purchased each 25 mg/ml dose for $2,900. In 2025, the price dropped to $2,700 and now, in 2026, to $1,440according to data from the Integrated Public Procurement System (Sicop).
In it private sectorAt the national level, each dose can be around $5,000.
This unaffordable price causes, among other factors, that the pembrolizumab is the medicine that is most demanded in the Constitutional Chamber. It represents one in four appeals for protections on medicines.
This report on this topic is part of the global investigation, The million dollar medicine (The Cancer Calculus)coordinated by the International Consortium of Investigative Journalists (ICIJ).
What did the CCSS commit to?
The CCSS achieved a 49.6% reduction in the price of the doses (up to $1,440) in a matter of two years and, in return, committed to expanding the number of tumors that will be treated with this drug. Now, will serve 21 categories and, consequently, also will increase the number of doses acquired.
This is how he explained it Esteban Vega de O, Logistics Manager of the Cajawho explained that multiple meetings and several counterproposals were required to reach an agreement with lto the firm MSD Central America Services, through which Merck markets its products in Central America.
Bonuses were requested
“We defined a proposal of how much we would be willing to pay and began a negotiation process. The company, of course, defines a price,” indicated the institution’s Logistics Manager.
“At that moment, we began to make counterproposals in the sense of what (quantity) can be subsidized. That is, if I am going to consume 100 bottles, give me 70 subsidized ones at zero cost and I pay the other 30. But, additionally, it makes me an improvement in the price of those remaining 30 with respect to the price I had been paying.
“It’s not that the company comes and says: ‘I’ll offer you this price’. No, no, that’s why we spent almost two years in that negotiation process, until we obtained an offer on the direct price of the product and on a quantity of discounted product that satisfied our needs and the capacity that we could cover and pay, without representing a financial risk for the institution,” said Vega de la O.
The official did not delve into the details of the agreement reached with MSD Central America Services. He specified that the company requested confidentialityclaiming that “prices are sensitive information” and that such a clause “constitutes a key element to achieve innovative access agreements in the countries.”
In the Sicop, it appears that the CCSS intends to acquire throughout this year, until December 15, 24,042 doses, for which it has a budget of up to $34.6 million.
Even in the first three months of the year, it already placed orders for a total of 9,500 doses, which together add up to $13.7 million.
The number of doses in the first quarter of 2026 is equivalent to the total acquisitions in 2025, with the difference that $25.7 million was invested last year, 47% more.
However, Vega de la O maintained that the amount spent last year on Keytruda was $31.2 million.
“Only in 2025, of all the savings achieved in the purchase of medicines, pembrolizumab represented a savings of nearly $9.8 million“, stressed the official of the Fund.
Company speaks out on ‘multi-year innovative agreement’
MSD Central America Services, through an email, also did not refer to what was agreed with the CCSS in a confidential manner. Solo insisted that it is committed to ensuring its products are “affordable” and promoting “health equity.”
“In 2025, we signed an innovative multi-year agreement with the CCSS for various types of cancer at different stages, providing 100% coverage and reimbursement of Keytruda in the public sector, for the indications approved as of December 2025,” added the company based in Panama.

(Design/The Nation/Design/The Nation)
For 21 tumors
Esteban Vega de O, Logistics Manager of the Cajainsisted that with the fall in price and the consequent lower weight within the institution’s finances, decided to expand the use of this immunotherapy treatment to 21 different types of tumors.
In addition to being prescribed for melanoma, Hodgkin lymphoma and cancers such as triple negative breast cancer, cervix, head and neckwill also be prescribed, without having to go to Room IV, to patients with kidney, gastric, lung, bladder, esophageal and gastroesophageal junction cancer.
“The authorization of use was approved by the Central Pharmacotherapy Committee for 21 different types of tumors, in which scientific evidence supports that this medication is truly a wonder for the control of the disease and is an immunotherapy that, depending on the type of cancer, especially breast cancer, can be curative.
”Costa Rica is the first country in Latin America to support the use of immunotherapy with pembrolizumab in 21 different types of tumors.. This was achieved through a negotiation process that generated significant savings of millions of dollars for the institution versus the price we had,” Vega de la O stressed.
Some patients die while waiting for the medicine
By increasing the number of tumors to be treated with Keytruda, the CCSS hopes to reduce the amount of protection resources they receive to deliver that medication.
That would help avoid the drama that dozens of patients go through to access this medication, who, on average, take up to six months for the prescription to be authorized.
That is the period that runs from when the specialist prescribes the medication, the Central Pharmacotherapy Committee denies it and Chamber IV orders its delivery.
In that period, some patients die while waiting for the medicine. The CCSS does not prescribe it for the first time due to its high cost and patients cannot buy it in the private sector either.
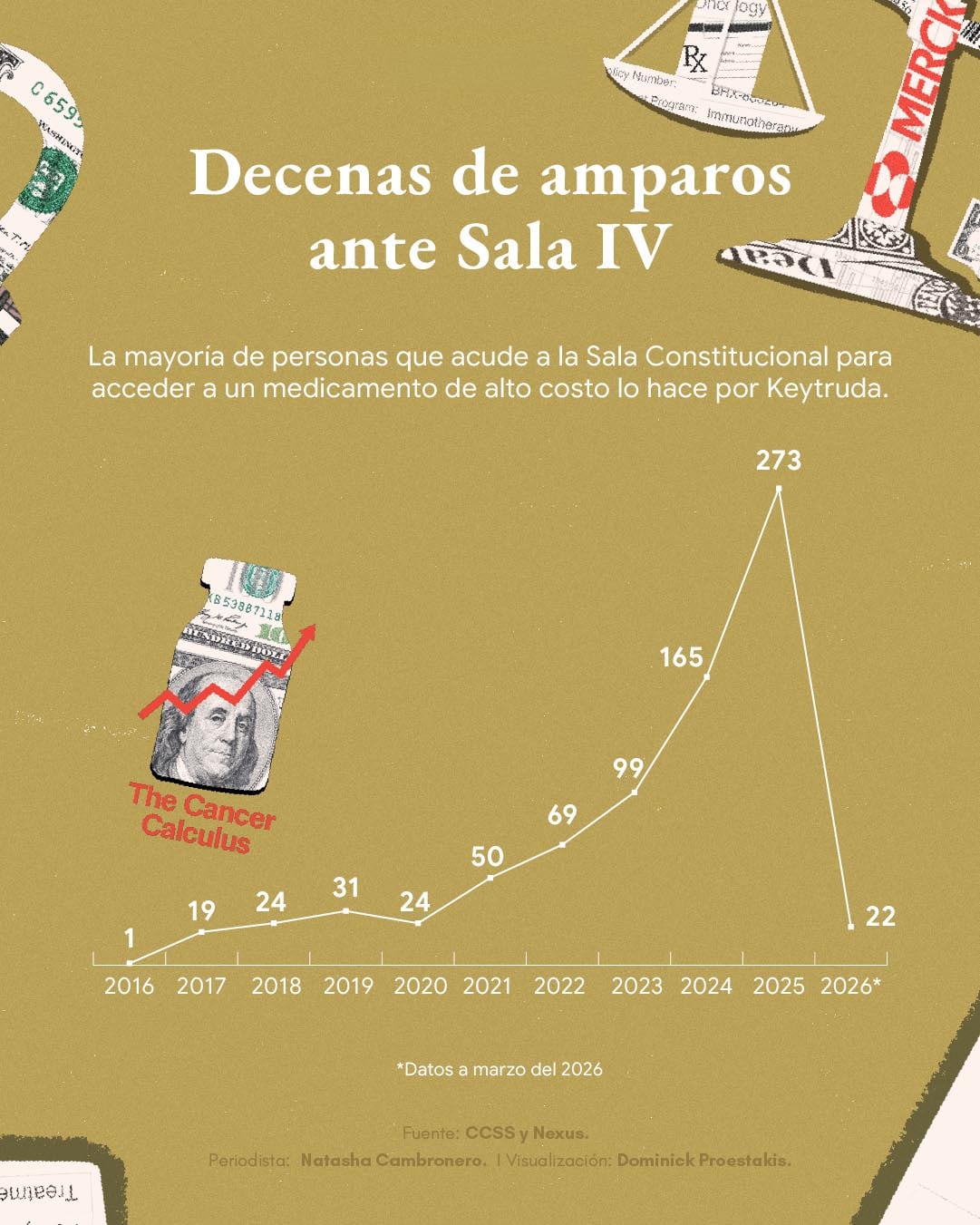
In 2024, the Fund received 648 appeals for protection due to access to high-cost medications and of that number, 165 (25%) were for pembrolizumab.
In second place was nintedanib – which is used to treat pulmonary fibrosis – with 48 protections, that is, 7.4%. The remaining 435 were for 54 other medications, according to data provided by Social Security.
Last year, the trend was maintained. On the Nexus platform, for jurisprudence of the Judiciary, 273 rulings from the Constitutional Chamber linked to Keytruda appear. And in the first three months of 2026 there are 22.
“With the aim of facilitating the expansion of its use at the institutional level, pembrolizumab was formally incorporated into the Official List of Medicines (LOM). This inclusion was finalized by agreement of the Central Pharmacotherapy Committee in December 2025,” stressed Vega de la O.
Chamber IV opts for third opinion
He president of the Constitutional Chamber, Fernando Castillospecified that, in general terms, the Court lasts, on average, from a month and a half to two months to resolve this type of protection.
He explained that, for the magistrates, the criteria of the prescribing doctor is not absolute nor is the criteria of the Central Pharmacotherapy Committee of the CCSS. Therefore, to resolve the clash of criteria, they ask for a third opinion, a expert opinion, to the Department of Forensic Medicine of the Judicial Branch.
And, if that unit does not have a suitable specialist, they turn to an expert designated by the board of directors of the College of Physicians and Surgeons.
“If this third criterion is favorable to the patient, then we declare in favor of the protection,” said Judge Castillo, who specified that 95% of the medications claimed are to treat some type of cancer and are high cost.
“In this type of analysis, the Court does not determine whether it is a specific pharmaceutical company. What we see is the legal controversy,” he added, while explaining that the third opinion does not consist of a reading of the file, but rather that the patient is summoned for an in-person check-up.
“The patient undergoes an examination as appropriate and in addition to that, the experts study the medical record and from there they provide a specific report,” Castillo stated.